The empirical formula mass of a covalent compound may be compared to the compound’s molecular or molar mass to derive a molecular formula. A compound’s percent composition provides the mass percentage of each element in the compound, and it is often experimentally determined and used to derive the compound’s empirical formula. The chemical identity of a substance is defined by the types and relative numbers of atoms composing its fundamental entities (molecules in the case of covalent compounds, ions in the case of ionic compounds). The molar mass is approximately 194 g/mol. 2 The molar mass of Oxygen is 15.999 g/mol. 1 The molar mass of Sulfur is 32.06 g/mol. You can see that The molar mass of Sodium is 22.990 g/mol. The mass % composition is as follows: C = 49.47% H = 5.191% N = 28.86% O = 16.48 %. So let’s look at the molar mass of Sodium, Sulfur and Oxygen from the above periodic table. Molar mass of molecular formula / Molar mass empirical formula = 90.3 g/mol / 46.0055 g/mol = 1.963 (to 3 SF) = 2Ĭaffeine contains hydrogen, carbon, nitrogen and oxygen. The given mass of K (4.7 g) is a bit more than one-tenth the molar mass (39.10 g), so a reasonable ballpark estimate of the number of moles would be slightly greater than 0.1 mol. 207.2 Pb Lead 114 (289) Uuq Ununquadium 10 20.1797 Ne Neon 18 39.948 Ar Argon 36 83.798 Kr Krypton 54 131. Then, we need to use the molar mass values to determine the molecular formula:Įmpirical formula molar mass = 14.0067 g/mol + 2(15.9994 g/mol) = 46.0055 g/mol Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. Calc4Chem is a JavaScript scientific calculator for chemists. to 2 sig fig’s) is close enough to round to the whole number, or NO 2 The corresponding numbers of atoms (in moles) are: 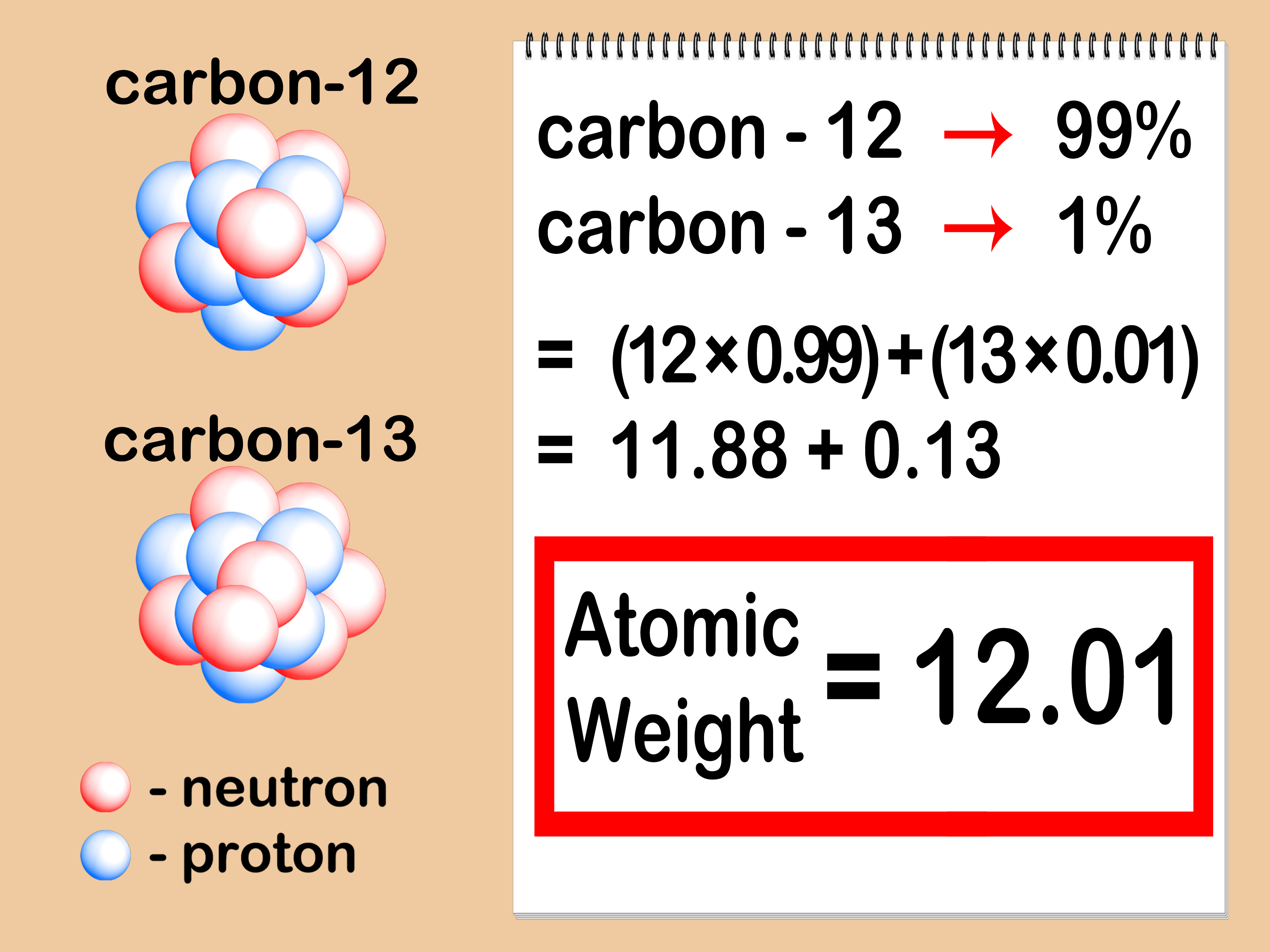
Consider a sample of compound determined to contain 1.71 g C and 0.287 g H. keep at least one decimal place in atomic masses from the periodic table. Calculate the molar mass of each compound. We then consider the moles of each element relative to each other, converting these numbers into a whole-number ratio that can be used to derive the empirical formula of the substance. Transition Metals and Coordination Compounds 3h 14m. To accomplish this, we can use molar masses to convert the mass of each element to a number of moles. Therefore, any experimentally derived data involving mass must be used to derive the corresponding numbers of atoms in the compound. However, we must keep in mind that chemical formulas represent the relative numbers, not masses, of atoms in the substance. NH3: 17.04 g/mol Mg(OH)2: 58.33 g/mol Fe2O3: 159.70 g/mol MOLAR MASS: The molar mass of a compound is the sum of all the atomic masses of the elements that constitutes the compound. Thus, the empirical formula = C 12/2H 4/2Cl 4/2O 2/2= C 6H 2Cl 2Oĭetermine the empirical formula for the following compounds:Ī) C 3H 8N b) CCl 4 c) C 2H 5 Determination of Empirical FormulasĪs previously mentioned, the most common approach to determining a compound’s chemical formula is to first measure the masses of its constituent elements. The molar mass of the following compounds in two decimal places are as follows. In a mole ratio problem, the given substance, expressed in moles, is written first.Determine the empirical formula for dioxin (C 12H 4Cl 4O 2), a very powerful poison. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
